 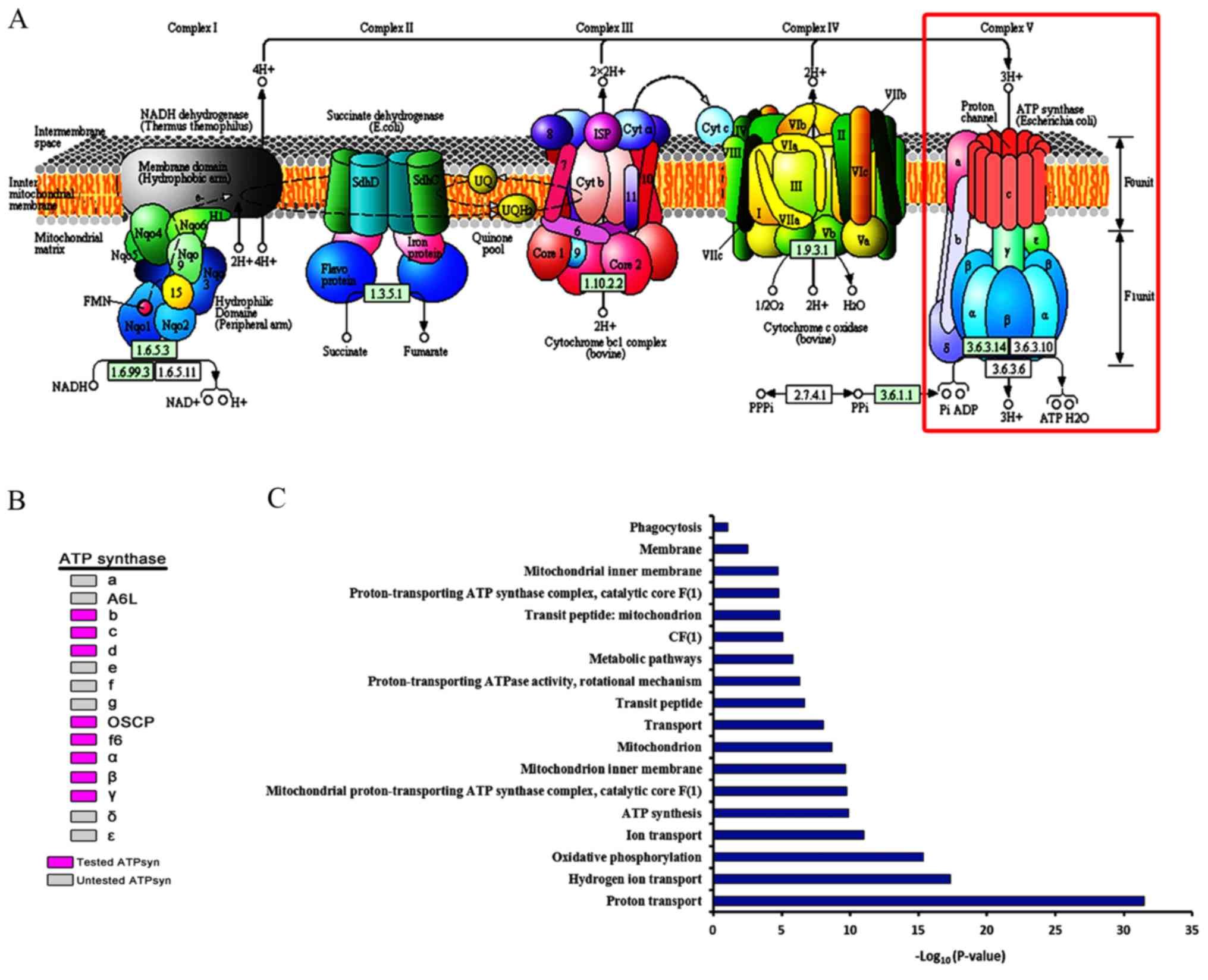 
In the first experiment (shown here), isolated organelles were placed in a medium with a low H + concentration until equilibrium was reached. Precisely how this mechanism uses ATP synthase to couple proton diffusion to ATP synthesis was demonstrated by two insightful experiments. 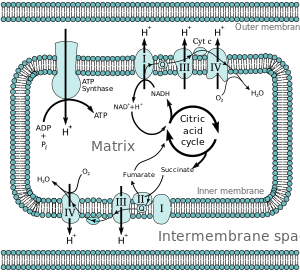
Previous research into the force driving ATP synthesis in mitochondria and chloroplasts led to the postulation of the chemiosmotic mechanism. Jagendorf’s experiment demonstrated that:ĪTP synthesis can occur even if light is not present.ĪTP synthesis can occur when thylakoids are suspended in buffer of pH 4.0.ĪTP synthesis in thylakoid can be promoted by addition of KOH.Figure 6.6 An Experiment Demonstrates the Chemiosmotic Mechanism The “stator” component of F 1−F o particles consists of:ģ α- and 3 β-subunits, δ-subunit, b-subunit of F 1 attached to “a” of F o They allow the movement of other cations.Įnergy released during proton movement in mitochondrial membrane in response to PMF is required by ATP synthase:įor binding of ADP and P i to β-subunits of F 1 They interfere with electron transport as a result proton gradient is not formed. They make the membrane permeable to protons. Their acidic nature combines with protons on the acidic side and diffuses to the other side of the membrane. Protonophores are the uncouplers which dissipate proton gradient due to the following: Interfere proton movement across membrane.Įlectron transport occurs but interfere with proton movement.Įlectron transport occurs but proton gradient is dissipated. Interfere with electron transport across ETC. Proton motive force across the membrane is created due to: Proton gradient is created by electron transport chain either during oxidation of NADH/FADH 2 or during the light reaction of photosynthesis in mitochondria and chloroplasts, respectively. Another mechanism involves coupling of energy of proton gradient with the synthesis of ATP. One of the ways is known as substrate-level phosphorylation, in which high-energy bond of the substrate is hydrolyzed and the energy released is utilized for ATP synthesis. There are two ways by which ATP can be synthesized in a cell. Thus, synthesis of 1 mole of ATP requires around 50 kJ of energy. However, under cellular conditions, the free energy value is around −50 kJ.mol −1. The standard free energy of ATP hydrolysis is 30.5 kJ.mol −1. In 1948, Alexander Todd undertook chemical synthesis of ATP and deciphered its structure, for which he was awarded Nobel Prize in 1957. Lipmann was the first to use the squiggle (~) symbol and the term “high-energy bond” for the compounds having high phosphate group transfer potential. Later on, Lohmann shared Nobel Prize with Hans Krebs in 1953 for his work on citric acid cycle. 
In 1941, it was demonstrated by Fritz Albert Lipmann that ATP was the universal energy carrier of the cells. ATP was discovered in 1929 by German chemist Karl Lohmann. Structure of ATP synthase and the mechanism of ATP synthesis have remained conserved through evolution. ATP synthase catalyzes ATP synthesis during oxidative phosphorylation and photophosphorylation. Various energy-requiring processes of the cell include transport of ions and biomolecules across plasma membrane against concentration gradient, movement of substances within the cell, cell growth including cell division, anabolic reactions of the cell, and various other cellular processes. It is utilized for the endergonic reactions and various energy-requiring processes in the cell. Adenosine triphosphate (ATP) is the energy currency of the cell and is required in cell metabolism of all living beings including bacteria, plants, and animals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
